Professional lash lifting is far more than a routine aesthetic service; it is a sophisticated chemical procedure that fundamentally alters the internal architecture of the lasahes. For the modern technician, navigating the molecular shifts occurring within the lash cortex is essential for delivering predictable results. Understanding how formulations interact with hair biology allows you to transition from following a protocol to mastering the science of a healthy, consistent lift while avoiding the risks of irreversible structural damage.
The Anatomy of a Lash
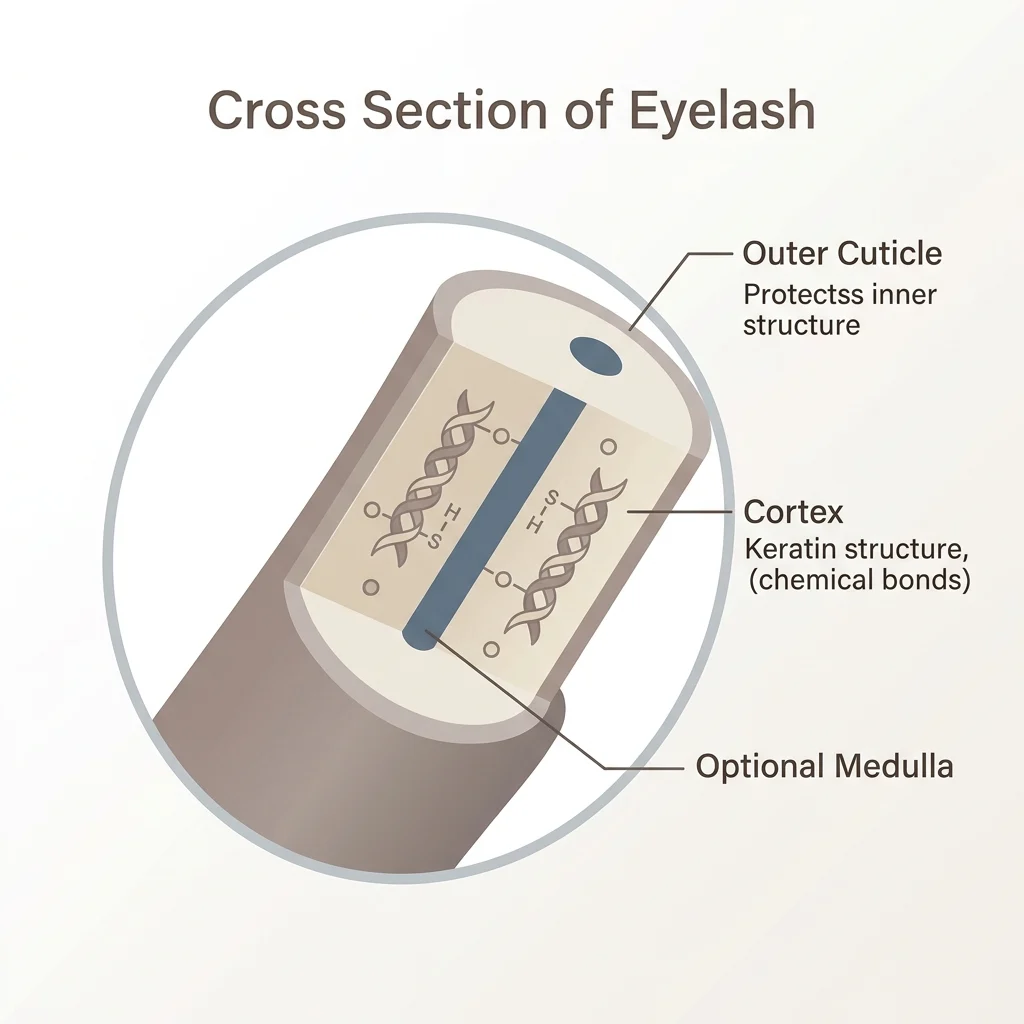
To understand the chemistry of the solutions, one must first look at the filamentous structure of the hair. Every eyelash is composed of a cuticle, a cortex, and occasionally a medulla. The cortex is the primary focus for chemical treatments, as it houses the helical keratin proteins that provide the lash with its inherent strength and shape. Within this protein structure, three distinct types of chemical bonds maintain the lash’s form.
Hydrogen bonds are temporary connections easily broken by water or heat, while ionic bonds, also known as salt bonds, are pH-dependent and respond to the acidity or alkalinity of the environment. The most critical, however, are the disulfide bonds. These are the strongest, most stable connections within the hair, and they must be chemically manipulated to achieve a permanent change in shape. A successful lash lift relies on a strategic two-step process to disrupt these lash chemical bonds and reform them in a new, lifted position against a silicone shield.
Step 1: The Chemistry of the Lifting Phase
The lifting solution, or Step 1, is formulated to swell the hair shaft and open the protective cuticle scales. This is achieved by creating a highly alkaline environment, typically maintaining a pH range between 8.5 and 9.5. Once the cuticle is lifted, the active reducing agents can penetrate the cortex to reach the disulfide bridges. During this reduction phase, the solution adds hydrogen atoms to the disulfide bonds, effectively “unlocking” the internal structure and making the hair malleable enough to take on the curve of the chosen silicone shield.
Reducing Agents: TGA vs. Cysteamine
Professional systems generally rely on one of two primary active reducing agents, each offering distinct advantages depending on the client’s lash health and texture. Ammonium Thioglycolate (TGA) is the traditional choice, known for its power and speed. It is particularly effective for coarse or resistant lashes, though it requires precise timing to prevent the hair from becoming over-processed.
Alternatively, many modern Korean lash lift systems utilize Cysteamine HCl. This ingredient is structurally similar to the hair’s own amino acids, allowing for a gentler interaction with the keratin structure. Cysteamine HCl-based systems often operate at a more controlled pH, helping to preserve the lash’s natural hydration and making them an ideal choice for clients with fine or sensitized lashes. To further protect the hair, high-quality lifting lotions are often enriched with nourishing buffers, which hydrate and calms the hair and skin throughout the chemical transition.
Step 2: The Neutralization and Fixing Phase
After the lashes have been sufficiently molded into their new shape, the chemical reaction must be stopped to prevent damage and lock the structure in place. This is the primary function of the Fixing Lotion, which acts as a neutralizer. Unlike the alkaline Step 1, the fixing solution is acidic, which works to neutralize the pH of the hair and close the cuticle.
The active ingredient in this step is typically Hydrogen Peroxide, which initiates an oxidation process. This process removes the extra hydrogen atoms added during the lifting phase, allowing the disulfide bonds to reform in their new, curved positions. For professional results, apply a consistent 1mm layer of fixing lotion to ensure uniform bond reformation. Technicians must strictly adhere to recommended processing times (such as keeping the lotion on the same amount of minutes as Step 1 lotion) to ensure the lift is stable and does not “drop” or lose its shape prematurely.
Step 3: Cuticle Closure and Nourishment
The chemical shifts required for a lash lift can leave the hair temporarily porous. To mitigate dryness, many professional systems include a final step designed for deep conditioning and protection. Utilizing advanced lash lift lotions for nourishment ensures that the internal cortex is shielded from environmental stressors.
Step 3 lamination creams act as sealants, closing the hair cuticles to lock in moisture. Formulations containing Hydrolyzed Keratin and Silk help rebuild the protein structure, while lipids from Argan and Jojoba oils restore the natural shine of the lashes. Applying such conditioner for 5 to 10 minutes at the end of the service results in lashes that are not only lifted but also appear thicker, darker, and more resilient.
Troubleshooting Chemical Interactions
Achieving the perfect lift requires balancing processing time with the porosity of the natural hair. Technicians must be vigilant against over-processed lashes, which occur when the lifting lotion remains on the hair for too long, causing the disulfide bonds to break too extensively. This results in a loss of structural integrity, manifesting as frizz or singed tips. Conversely, under-processing occurs when the reducing agent fails to break enough bonds, often due to low room temperatures or resistant, low-porosity hair types.
To maintain safety and excellence, always conduct a thorough consultation to identify contraindications and perform a patch test at least 24 to 48 hours before the service. Mastering these chemical nuances allows you to tailor every treatment to the specific needs of the client, whether they require a classical lash lift for dramatic results or a gentle Cysteamine HCl system for a more subtle, nourishing effect. Selecting the correct chemistry is the most effective way to guarantee the long-term health and integrity of your clients’ lashes.